“Are There Size Limitations For The SAPIEN Aortic Valve Replacement?” Asks Anita
By Adam Pick on February 21, 2012
I just received an interesting question from Anita about the SAPIEN heart valve replacement.
Anita wrote to me, “Hi Adam, My dad is not doing very well. At 86, he’s been diagnosed with severe aortic stenosis. We understand that he might be a candidate for the SAPIEN valve. My dad is a big guy. I’m curious to know if the valve can expand to fit all valve sizes. Or, are there limitations given the size of the catheter? By the way, what is the normal size of an aortic valve?”
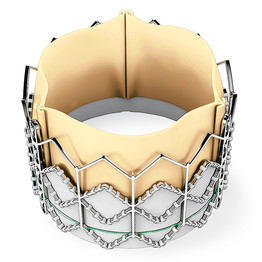
Edwards SAPIEN Aortic Valve Replacement
I wanted to provide Anita an expert opinion so I contacted Dr. Craig Smith, the principal investigator for the SAPIEN clinical trials.
In response to Anita’s question, Dr. Smith offered several insights. First, he addressed the available sizes and constraints of the SAPIEN aortic valve replacement:
The SAPIEN valve comes in two sizes (23 and 26 mm), with a 29 mm size that is just beginning to be used in the US. At least 80% of patients are treatable with the 23 or 26 valves. Relatively few “normal” valves are smaller than 18 mm or larger than 26 mm, as they are measured by echocardiography or CT scan. A “big” man doesn’t necessarily have a “big” valve, especially if “big” refers to weight more than height. There are size limitations for the valve delivery system based on the size of the major blood vessels in the lower body, but the transapical delivery alternative allows most patients to be treated regardless.
Dr. Smith then addresses the utility of the SAPIEN relative to other traditional, surgical approaches and devices:
If this man is in good general health for an 86-year-old, the SAPIEN valve might not be a relevant consideration, and he should have conventional open-heart aortic valve replacement (AVR). This will be true of one-third to one-half of patients over 80. If his surgical risk is moderately elevated by issues in addition to age, he would be eligible for the PARTNER II Trial of the SAPIEN valve, in which he would be randomized to receive either the SAPIEN valve or conventional aortic valve replacement (50% chance of receiving SAPIEN). If his surgical risk is extreme, as determined by one or more surgical opinions, he can receive the SAPIEN valve without being part of a trial and without being randomized. These options can be sorted out at any center participating in the SAPIEN trials. Similar trials of the CoreValve device (different design) are ongoing in earlier stages, and I’m sure any CoreValve center would be delighted to evaluate him as well.
Thanks to Anita for her question and a special thanks to Doctor Craig Smith, cardiac surgeon, for sharing his clinical experience with this new device.
Keep on tickin!
Adam
|
Mike Rehmus says on February 21st, 2012 at 8:46 pm |
|
My dad had an atrial valve replacement at age 87 and lived to age 93, not dying from issues with the valve or the replacement. He breezed through the surgery and recovery better than I have at age 69 so age isn’t the issue it used to be. I do wish him good luck in whatever solution he chooses. |
 |
|
Jackie Ellason says on March 11th, 2012 at 7:44 pm |
|
My 82 year old husband is scheduled to receive a 26 mm TAVR on March 20th. He is considered “too high risk” for open-heart replacement. The exact measurement of his valve was described as “difficult to see” on two separate TEE exams. He is a “big” man, 6’6″. We have been told the 29mm valve was not available in the US. Dr. Smith’s reply to Anita would seem to indicate otherwise. Please advise. Thank you. |
 |














