One Step Closer To FDA Approval For SAPIEN Aortic Valve?
Within our patient and caregiver community, there is great excitement about transcatheter aortic valve replacement technology. These non-invasive approaches for aortic stenosis treatment are often described with adjectives including revolutionary and transformational.
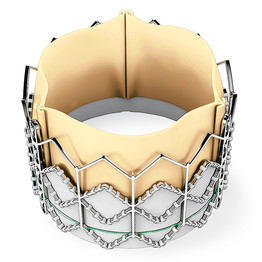
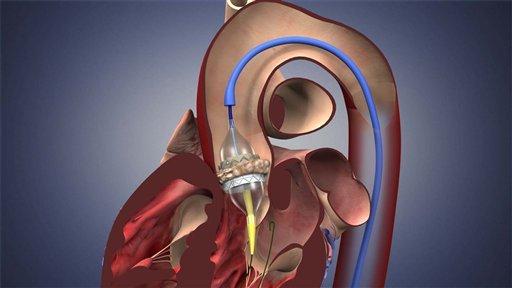
I wanted to relay new information about the FDA approval process for one of these percutaneous devices, the SAPIEN aortic valve replacement by Edwards Lifesciences. This valve, which does not require an incision to the sternum or ribs for implant, received two favorable votes from an advisory panel of cardiologists which reviewed its most recent clinical trial:
The panel voted 9 to 0 that the SAPIEN's benefits outweigh its risks (one member of the panel abstained).
The panel voted 7 to 3 that the SAPIEN is safe for its intended patient category, which is comprised of high-risk patients that are too frail for traditional surgical approaches.
This is a significant step for the SAPIEN as there continues to be concern about the increased rate of stroke and patient complications for SAPIEN trial recipients, according to Medical News Today.
Dr. Augusto Pichard, a principal investigator for the SAPIEN trial at the Washington Hospital Center, said that although vascular complications declined during the trial, the causes of a 7.3% stroke rate in the first month and 10.6% rate in the first twelve months were unclear. However, looking forward, Dr. Pichard called the potential approval a "saving grace" for inoperable patients with severe aortic stenosis. (To learn more about stenosis, click here.)
While this minimally invasive device -- which is delivered through the femoral artery or the left ventricle -- has yet to receive FDA approval in the United States, the SAPIEN is commercially available in Europe. (On Sunday, my dad told me that one of his friends just traveled to Germany to have the SAPIEN implanted.)
Interestingly, the FDA does not need to accept the view of its advisory panels. Several reports, however, suggest this is a common practice. As of yet, no date has been provided as to when the official FDA evaluation will come for the SAPIEN device.
Needless to say, I will continue to monitor the SAPIEN and other technologies that could greatly impact valvular treatment in the future.
Keep on tickin!
Adam
Written by Adam Pick
Patient & Website Founder

Written by Adam Pick - Patient & Website Founder
Adam Pick is a heart valve patient and author of The Patient's Guide To Heart Valve Surgery. In 2006, Adam founded HeartValveSurgery.com to educate and empower patients. This award-winning website has helped over 10 million people fight heart valve disease. Adam has been featured by the American Heart Association and Medical News Today.
Comments
Adam's Newest Blogs
New Community Posts
Blog Categories
- Adam's Updates
- Aneurysms
- Aortic Stenosis
- Aortic Valve Repair
- Aortic Valve Replacement
- Atrial Fibrillation
- Before Surgery
- Bicuspid Aortic Valve
- Heart Valve Replacement
- Medical Technology
- Mitral Regurgitation
- Mitral Valve Repair
- Patient Stories
- Pulmonary Valve
- Recovery
- Ross Procedure
- Surgeons & Hospitals
- Tricuspid Valve
Adam's Newest Blogs
Blog Categories
- Adam's Updates
- Aneurysms
- Aortic Stenosis
- Aortic Valve Repair
- Aortic Valve Replacement
- Atrial Fibrillation
- Before Surgery
- Bicuspid Aortic Valve
- Heart Valve Replacement
- Medical Technology
- Mitral Regurgitation
- Mitral Valve Repair
- Patient Stories
- Pulmonary Valve
- Recovery
- Ross Procedure
- Surgeons & Hospitals
- Tricuspid Valve
Blog Categories
- Adam's Updates
- Aneurysms
- Aortic Stenosis
- Aortic Valve Repair
- Aortic Valve Replacement
- Atrial Fibrillation
- Before Surgery
- Bicuspid Aortic Valve
- Heart Valve Replacement
- Medical Technology
- Mitral Regurgitation
- Mitral Valve Repair
- Patient Stories
- Pulmonary Valve
- Recovery
- Ross Procedure
- Surgeons & Hospitals
- Tricuspid Valve
New Posts From Our Community

Ercan Afacan from
Montreal, Canada
6 Months Post-op!! Completed Rehab. For those going through recovery, hang...
Meet Ercan
Heather Leigh from
Brooklyn, New York
Hi all!! Surgery went well- robotics worked so it was just...
Meet Heather
Find Heart Valve Surgeons
Search 1,500 patient-recommended surgeons