Breakthrough Innovation: The HAART Aortic Annuloplasty Device Cleared by the FDA
Written By: Adam Pick, Patient Advocate, Author & Website Founder
Page Last Updated: May 14, 2025
A renaissance is taking place in the world of aortic valve repair.
While patients have benefited from advances in mitral valve repair procedures (long-term durability, freedom from re-operation and normal life expectancy), reproducing those results using aortic valve repair techniques has been challenging.
Unfortunately, patients with aortic valve regurgitation – also known as aortic insufficiency – had limited treatment options, including tissue and mechanical heart valve replacement devices.
In case you missed it… The HAART Aortic Annuloplasty Device received clearance from the U.S. Food & Drug Administration (FDA). This is a very important development for patients who are searching for a potential long-term fix for their leaking aortic valves. The great news is that the early operative results using the HAART device have supported a global trend toward aortic valve repair.
Surgeons Say This About Aortic Valve Repair & The HAART Device
To learn more about this exciting innovation, I invited four world-renowned heart surgeons to answer seven important questions about their experiences with aortic valve repair and the HAART device.
Our panel includes:
Dr. Vinay Badhwar - Gordon F. Murray Professor and Chair, Department of Cardiovascular and Thoracic Surgery, West Virginia Universty and Chair of the WVU Heart and Vascular Institute.
Dr. Marc Gerdisch - Chief of Cardiovascular and Thoracic Surgery at Franciscan Health in Indianapolis, Indiana.
Dr. S. Chris Malaisrie - Associate Professor of Surgery (Cardiac Surgery) at Northwestern Medicine and the Associate Director of the Bluhm Cardiovascular Institute’s Center for Heart Valve Disease and Co-Director of its Thoracic Aortic Surgery Program in Chicago Illinois.
Dr. Ming-Sing Si - Surgical Director of Pediatric Heart Transplantation and Adult Congenital Heart Surgery at the Michigan Medicine Congenital Heart Center in Ann Arbor, Michigan.
1. What are the current treatment options for patients with aortic valve regurgitation?
Dr. Gerdisch: “Aortic valve regurgitation or insufficiency has many forms, depending on the underlying mechanism. When it is not associated with too much bulky calcium or destruction of the valve leaflets, aortic regurgitation can be repaired. The extent of involvement of the aortic valve leaflets and the first portion of the aorta, where the valve is housed, will determine the complexity of the repair. When the valve is not repairable or the patient and surgeon decide not to pursue repair, the valve can be replaced. There are various replacement valves, all of which have a role and serve great purpose, but all of which share risks that the patient’s native valve does not convey.”
Dr. Si: “Surgery can be classified as replacement or repair. Replacement options can be further divided into mechanical prosthesis, bio-prosthesis (open or transcatheter placed), or autograft (Ross procedure). A new replacement method is just leaflet replacement (Ozaki procedure).”
2. What are the advantages of aortic valve repair compared to aortic valve replacement?
Dr. Malaisrie: “While valve replacement options have improved, artificial valves still have the disadvantage of long-term deterioration (tissue valve) and the need for lifelong anticoagulation (mechanical valve). Valve repair offers restored function of the patient’s own native valve, avoiding these disadvantages.”
Dr. Gerdisch: “Replacement prosthetic valves provide alleviation of the mechanical problem from which the heart is suffering due to the diseased valve, and as such, extend and save lives. Nonetheless, changing a person’s valve for a prosthetic valve is in a sense, ‘trading diseases.’ All prosthetic valves, whether mechanical or tissue are vulnerable to clot forming on the surface, and the possibility of embolization and stroke. The risk is small, but present over the entirety of the recipient’s life. Furthermore, tissue valves deteriorate over time and the speed of deterioration is increased for younger patients. Mechanical valves require lifelong anticoagulation. These factors need to be weighed in designing an algorithm to address a person’s valve, both with respect to timing and repair versus replacement.”
Dr. Gerdisch continues, “Aortic valve repair provides patients with the opportunity to keep their native tissue and avoid the issues described above. However, not every insufficient valve can be repaired, and not every repaired valve will last the rest of the patient’s life. Indeed, some patients may need a second operation someday, to replace the valve. However, the years spent with the repaired valve lack the risks intrinsic to prosthetic valves.”
Dr. Badhwar: “The advantage of aortic valve repair is the patient’s native anatomy and the avoidance of prosthesis-related complications such as structural valve degeneration, thrombosis and oral anticoagulation.”
Dr. Si: “When aortic valve repair is compared to mechanical replacement, there is no need for anticoagulation, possible decreased infectious risk, possible increased durability, and possible improved survival. When aortic valve repair is compared to biological tissue prosthesis: possible decreased infectious risk, possible increased durability, and possible improved survival.”
3. How are aortic valve repair procedures performed?
Dr. Si: “Aortic valve repair is performed as an open heart surgery. The procedures can be performed via a median sternotomy or minimally invasive incision.”
Dr. Malaisrie: “Valve repair consists of two parts. The first is leaflet repair mostly to address prolapse (a floppy valve). The second part of the operation is annulus repair which addresses the frame of the aortic valve.”
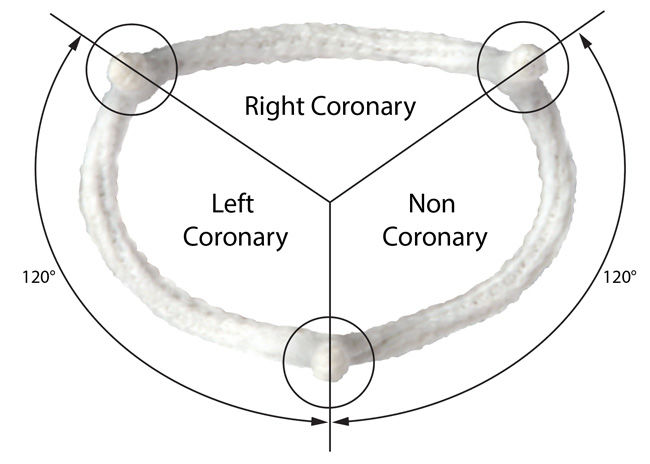
Dr. Gerdisch: “There are various methods of aortic valve repair. All attempt to restore the geometry of the valve and the overlap of the leaflets, to eliminate the leak and reduce the stress on the leaflets, allowing them to resume normal function. As mentioned above, often aortic valve repair is combined with replacement of part or all the ascending aorta due to associated aortic aneurysm. All repair techniques share the fundamental principle of trying to establish the correct dimensions for the aortic annulus. Once that has been achieved, the surgeon can set about making fine adjustments to the leaflets to arrive at a well-functioning valve.”
4. What devices do you use to complete an aortic valve repair?
Dr. Gerdisch: “The HAART 200 and 300 annuloplasty rings provide a reproducible means of creating normal 3-dimensional aortic annulus architecture. The aortic annulus has its own anatomy, with specific 3-dimensional geometry. Using the word “ring” for these devices is a dramatic oversimplification. The HAART rings were developed from scans made of normal humans, translating the consistent geometric pattern into three dimensional rings that are implanted below the aortic valve, dictating the shape of the annulus, and the relationship of the leaflets. The pathologic changes in the annulus configuration are remedied, and the leaflets may then be altered using fine suture to perfect their contour. Rings have been designed for trileaflet (normal) and bicuspid valves.”=
Dr. Malaisrie: “Several annulus repair devices are available. But, not all are available in the US. I use either a Dacron graft or an intra-annular ring (HAART ring) to complete the repair.”
Dr. Si: “I use the HAART device to stabilize the annulus and then perform leaflet repair. If the root is enlarged, I would replace that as well.”
To help you learn how the HAART devices are implanted, here’s a great video animation:
5. How are your patients doing who received the HAART?
Dr. Badhwar: “Excellent. Clinically as well as echocardiographically at 30 days and 1 year, patients all have restorative anatomy with less than mild aortic insufficiency without need of re-intervention.”
Dr. Gerdisch: “Patients for whom we have performed aortic valve repair with the HAART devices have done very well. Like any reconstructive operations there will be limitations due to very advanced pathology or tissue abnormalities but the technique has seen dramatically broader application. As we have developed a deeper understanding of the relationship of the aortic valve leaflets, the annulus and the composite of the aortic root, the procedure is being refined and extended to more complex scenarios of extensive aortic surgery and multi-valve operations.”
Dr. Malaisrie: “Patients are all alive and doing well.”
Dr. Si: “The experience is very early, but the patients are all doing well.”
6. What is the future of aortic valve repair? Will it become the new standard for patients with leaking aortic valves?
Dr. Si: “I think the future is bright. Aortic valve repair is starting to become more widely practiced as more surgeons become trained in these techniques.”
Dr. Badhwar: “In the future, we will see more applicability of adjunctive techniques to be able to address increasing complexity. Surgical replacement of central aortic insufficiency with trileaflet or bicuspid anatomy can be readily managed with the trileaflet or bicuspid annuloplasty ring. With this device, the technical advancement to address fused raphe, rheumatic disease and complex prolapse are all possible.”
Dr. Malaisrie: “Devices to repair the annulus of the aortic valve is a major step forward. Time will tell which devices provide the most durable outcomes.”
Dr. Gerdisch: “As we move toward more standardized methods of aortic valve repair, I expect progressive adoption by surgeons. As is always true with combinations of device and technique evolution, it will expand slowly, as surgeons develop the skills to perform aortic valve repair effectively and safely. With additional long-term follow-up and surgeon adoption, it will enter the conversation about treatment of aortic insufficiency more frequently and allow patients and doctors to have a thorough discussion, about the treatment option that will best suit their needs and expectations.”
7. What advice do you have for aortic regurgitation or aortic insufficiency patients wanting an aortic valve repair?
Dr. Gerdisch: “People with leaking aortic valves should devote time to researching options and discussing the opportunities for treatment. If they would like to be considered for aortic valve repair, they should visit with an aortic valve repair surgeon. Decision-making about aortic regurgitation is complex and requires assessment of not only the valve, but the entire patient. Although, ultimately the patient and surgeon need to meet in-person, often the surgeon can review records and images that are sent and simply share the findings with the patient by phone. At least then the patient learns about the potential for the valve to be repaired.”
Dr. Si: “They should discuss this option with their surgeon (who is able to perform repair procedures) in detail.”
Keep Learning About The HAART Innovation!
I hope this helped you learn more about the use of the HAART device to treat aortic regurgitation.
I would like to thank each of the esteemed surgeons – Drs. Badhwar, Gerdisch, Malaisrie and Si – for sharing their research and clinical experiences specific to aortic valve repair! If you have questions about the HAART Aortic Annuloplasty Devices, I’d encourage you to speak with your doctor.
Related Link:
Keep on tickin!
Adam
Written by Adam Pick
Patient & Website Founder

Written by Adam Pick - Patient & Website Founder
Adam Pick is a heart valve patient and author of The Patient's Guide To Heart Valve Surgery. In 2006, Adam founded HeartValveSurgery.com to educate and empower patients. This award-winning website has helped over 10 million people fight heart valve disease. Adam has been featured by the American Heart Association and Medical News Today.
Upcoming Surgeries
Surgeon Spotlight
Dr. Christopher Heid is a leading cardiac surgeon at UT Southwestern in Dallas, Texas who specializes in heart valve repair and replacement operations.